 This is the third post in a five-part series where we'll give you a look inside this important new COVID-19 resource. In case you missed it, we recommend viewing the series in order, starting with Part One.
This is the third post in a five-part series where we'll give you a look inside this important new COVID-19 resource. In case you missed it, we recommend viewing the series in order, starting with Part One.
As an EMS provider, staying safe through the evolving COVID-19 situation involves distinct challenges. That's why the Public Safety Group, in partnership with the American Academy of Orthopaedic Surgeons, is pleased to introduce Evolution of EMS: COVID-19 Guidance for EMS Providers. This unique resource offers strategies and insights designed specifically for you.
This excerpt looks at the vaccine development process and how the process for a COVID-19 vaccine differs, and provides a survey of promising vaccine candidate types.
Evolution of EMS: COVID-19 Guidance for EMS Providers describes the SARS-CoV-2 virus and disease, prevention tactics, vaccine development, treatment, and public health implications, particularly as they affect providers working in the field. It’s not designed to supply everything you need to know about COVID-19, but rather to brief you on key issues and considerations. COVID-19 Guidance for EMS Providers also offers references for learning more and staying up to date. The goal: empowering you to protect yourself, your co-workers, and those you serve more efficiently and effectively.
There are three easy ways to access COVID-19 Guidance for EMS Providers:
- Stay Tuned to the Public Safety Group Blog:
New excerpts will be rolled out on our Public Safety Group blog on a biweekly basis, free of charge (Part One is available here, Part Two is available here, Part Three is available below, and Parts Four and Five will be available in the coming weeks)
- Sign Up to Receive a Free, Complete Copy of the Resource:
Visit http://go.psglearning.com/COVIDGuidanceNow to request your free copy today. We'll email you a PDF to download as soon as the complete resource is available later this Fall:
I Want to Request My Free PDF:
COVID-19 Guidance for EMS Providers
- Purchase Physical Copies of the Resource on our Website:
Visit http://go.psglearning.com/COVIDGuidance to pre-order your physical copy which will be available later this Fall
Questions? Please contact your dedicated Public Safety Specialist today.
Part Three: Vaccine Development
Since the outbreak of COVID-19, scientists and vaccine specialists have worked to unlock the genetic code of the SARS-CoV-2 virus and develop an effective vaccine. By September 2020, more than 100 vaccine candidates were in development throughout the world, and WHO reported that 40 had advanced to clinical trials.
As an EMS provider, you could be among the first to receive a COVID-19 vaccine and may be tasked with administering the vaccine to the community (Figure 3-1). Reviewing the course of vaccine development, and understanding the range of candidates being tested, will help prepare you to implement your agency’s policies and make sound decisions about your own health.
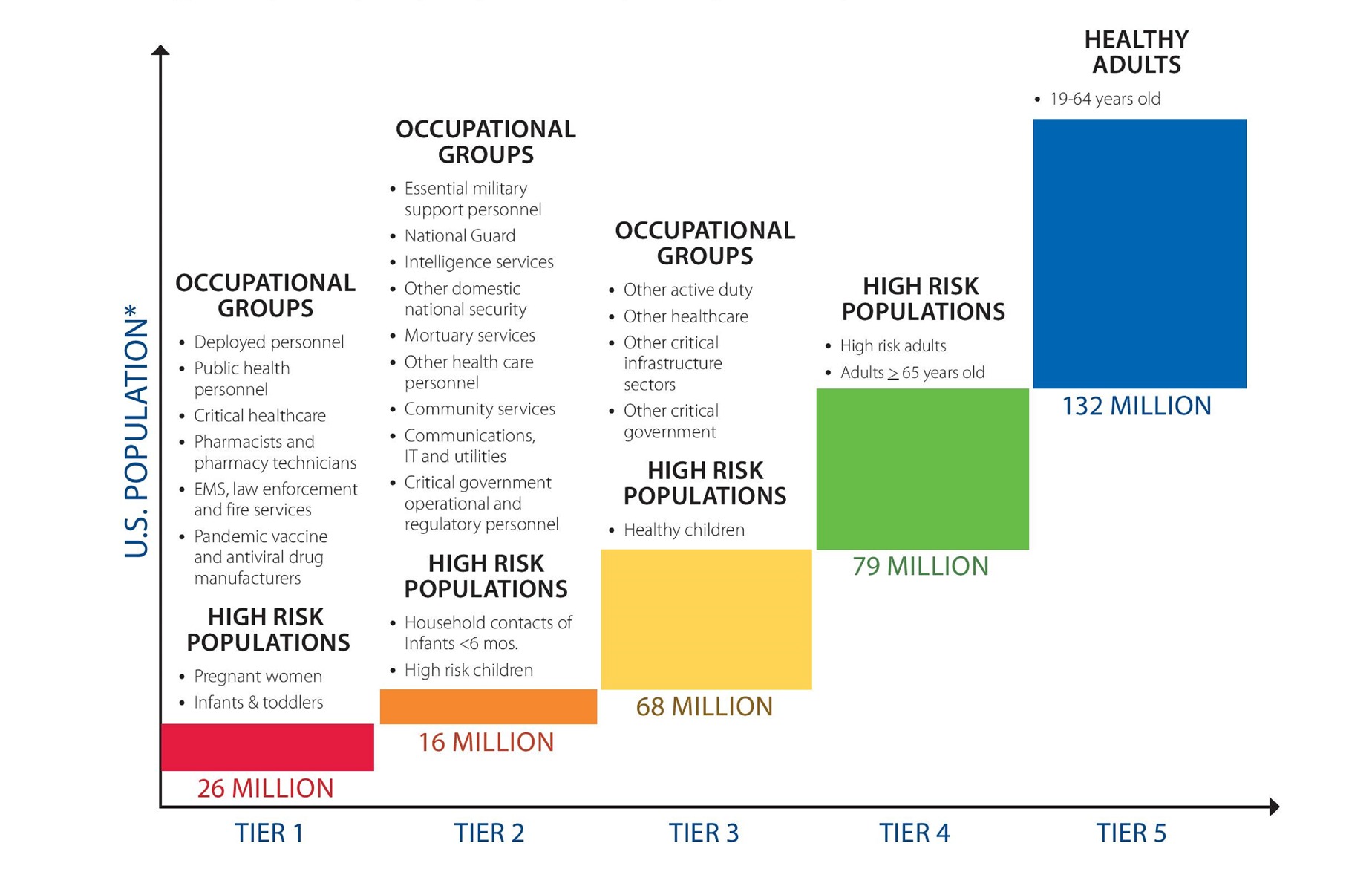
Figure 3-1: Tiered vaccine administration structure for high or very high pandemic severity.
Reproduced from Centers for Disease Control and Prevention. 2018. “Allocating and Targeting Pandemic Influenza Vaccine During an Influenza Pandemic.” US Department of Health and Human Services. Accessed October 8, 2020.
*Based on 2015 US population of 321 million people.
Typical Course of Development
Developing a COVID-19 vaccine involves the same basic steps used when developing other vaccines:
- Exploratory stage
- Preclinical stage
- Clinical development
- Regulatory review and approval
- Manufacturing
- Quality control
As a solution to the global pandemic, steps are being taken to expedite the COVID-19 vaccine process (Figure 3-2). In the United States, this expedited process requires condensing and overlapping some phases while maintaining strict review and quality control by the FDA.
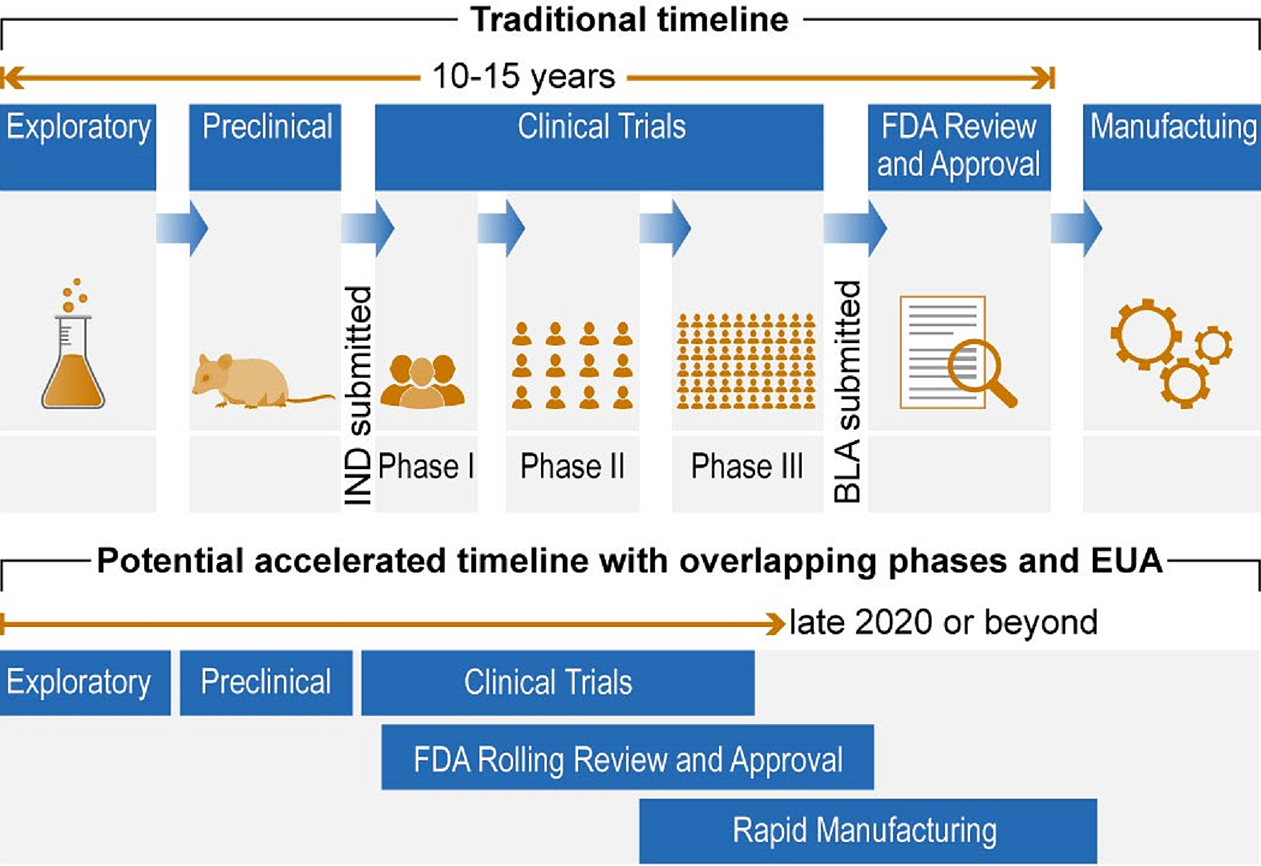
Figure 3-2: Timelines for vaccine development.
Government Accountability Office. 2020. Science & Tech Spotlight: COVID-19 Vaccine Development—GAO-20-583SP. May 26, 2020.
*Abbreviations: BLA, Biologics License application; EUA, Emergency Use Authorization; FDA, Food and Drug Administration; IND, Investigational New Drug
Exploratory and Preclinical Stages
These initial stages involve lab research and animal studies. If a virus is known to scientists, the objective after genome sequencing is to determine how to get the human body to build immunity to the disease without suffering its symptoms. For example, with a typical intranasal flu vaccine, an attenuated virus—one that has been genetically altered so it cannot cause disease—is administered. Once exposed, the body elicits an immune response in the local tissue, and antibodies are built against the virus that circulate through the body.
Other vaccines take a surface protein, a specific protein found on the virus and unique to the virus, and expose the immune system so antibodies develop against that protein. Because that protein is located only on the virus that causes the disease, the body builds immunity and destroys that protein if the immune system recognizes it, thus eliminating the virus.
If the virus is unknown to scientists, this exploratory process can be much more tedious, as the virus has to be safely replicated and analyzed prior to determining which type of vaccine may be most effective. Typically this process can last 3 to 6 years. Once a vaccine has been developed in the lab and safely tested on animals, it reaches clinical development and is ready for human testing.
Clinical Development
In the United States, all vaccines must go through a rigorous approval process to ensure they are safe and effective, and that the benefits outweigh the risks. Typically, this stage of development involves three phases (Figure 3-3).
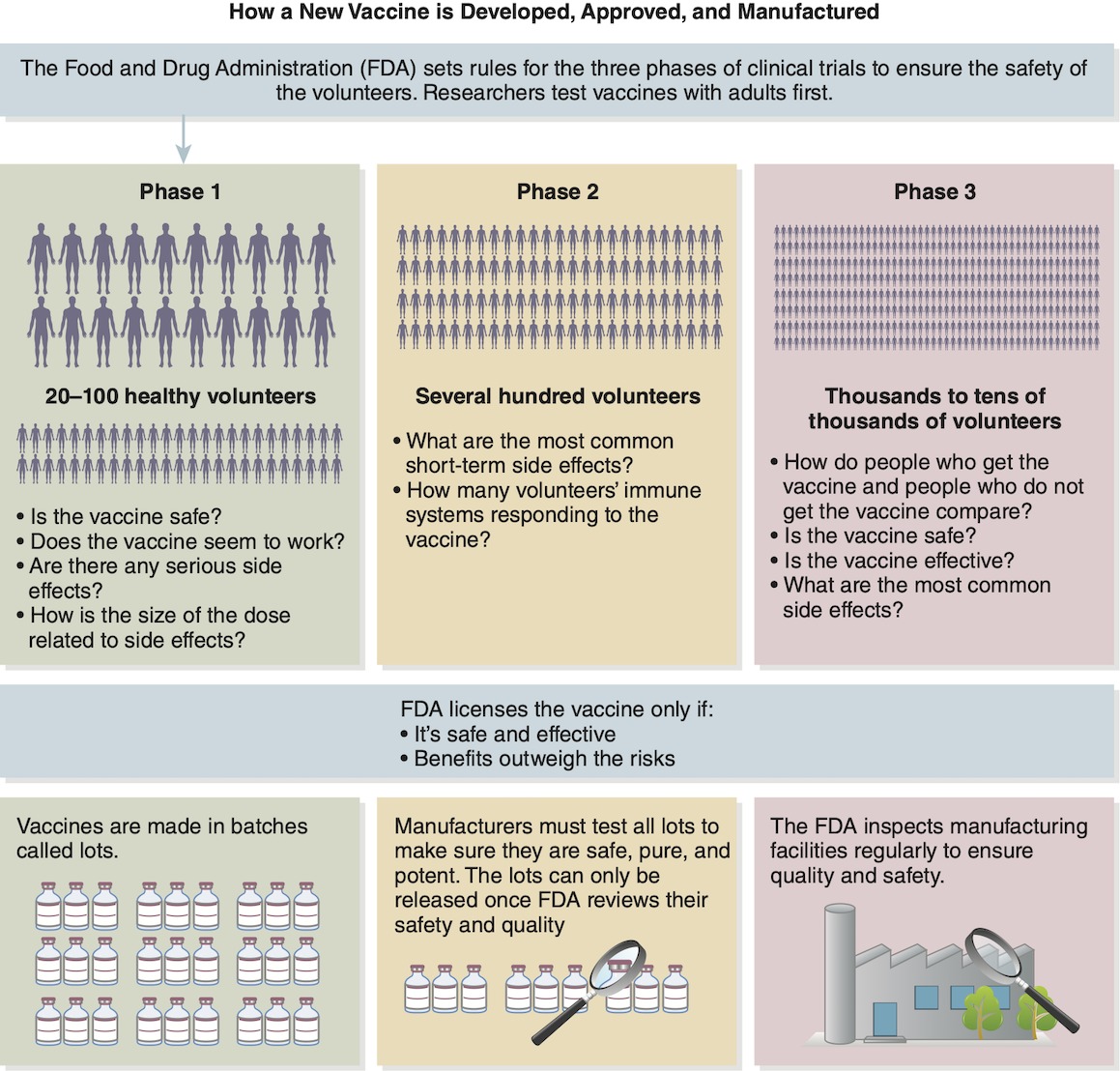
Figure 3-3: Typical testing process for a new vaccine.
Modified from Centers for Disease Control and Prevention. 2018. Provider Resources for Vaccine Conversations With Parents: Ensuring the Safety of Vaccines in the United States. Accessed October 8, 2020.
Before official testing can begin, the FDA, which oversees vaccine testing, requires the vaccine to be inspected and licensed by its review committee. Following this initial review, phase 1 requires a vaccine to be administered to approximately 20 to 100 volunteers. Starting with a small group enables researchers to determine human response and side effects in a more controlled manner.
If a vaccine meets the criteria of phase 1, it moves on to phase 2, where it is given to several hundred volunteers. The vaccine is then evaluated for short-term side effects and immune system response. Typically, phases 1 and 2 take at least 3 months but may last years, as those who received the vaccine are monitored to compare them with patients who did not receive the vaccine or with those who received a placebo instead of the vaccine.
In phase 3, the pool of recipients widens yet again, typically to tens of thousands of people. The participants are monitored to determine the vaccine’s effectiveness and side effects and to compare the vaccine recipients with a control group who received a placebo. This final phase can take several months to several years.
Review and Approval
Following successful completion of the trial phases by a vaccine’s development groups, each country can elect to approve it for human use. In the United States, review and approval involve rigorous oversight by the FDA and continued monitoring after widespread use. You likely have heard of drugs being recalled or taken off the market because of consequences not fully explored in the testing phase, or only recognized after years of use. Although this is rare, it does occur.
Depending on its internal policies and regulations, a country may choose to expedite testing or skip certain phases of testing altogether. Although the COVID-19 vaccine may well be the fastest vaccine to the hit the market, there are some countries that would like to make vaccine availability even faster. In fact, multiple news reports suggest that, based on favorable phase 2 trials of two different COVID-19 vaccines, China will allow vaccinations to be given to some military personnel and government employees prior to the completion of phase 3 (Wee and Simões 2020). Other reports suggest Russia also may provide a China-based vaccine to citizens prior to the completion of phase 3 testing.
Conflicting reports on vaccine use in these and other countries continue to emerge. What is clear is that solutions to the pandemic are weighed against the health of the public and the safety of the vaccine and can vary based on culture, tradition, and government policy (Callaway 2020).
Manufacturing and Quality Control
After the review process is complete and government agencies approve the new vaccine, it can be manufactured and distributed to licensed facilities. This may sound like a quick part of the process, but manufacturing a sufficient quantity of the medium, syringes, needles, and packaging—all under safety control—takes time. Pharmaceutical companies typically have years to prepare the supply chain for a new vaccine, yet the timelines proposed for the COVID-19 vaccine require mere months.
After preparation and shipment, which will likely occur in batches, the public must be vaccinated. Although most of the population will obtain the vaccine from their health care provider, some citizens will require more convincing. As a health care provider, you may be looking forward to getting vaccinated, but a poll conducted in July 2020 indicated that only 65% of Americans would get a free, FDA-approved COVID-19 vaccine if it were available (O’Keefe 2020). Thus, a public health campaign, similar to what occurs every year for the flu shot, will be essential, wherein health care officials and public figures advocate for immunization and explain its benefits.
Beating the Clock
Although vaccine development in the United States typically takes anywhere from 5 to 20 years, the Department of Health and Human Services (HHS) has responded to the COVID-19 pandemic by launching Operation Warp Speed. This private-public partnership set the goal of delivering 300 million doses of a safe and effective COVID-19 vaccine by January 2021. If this timeline holds, this would be the fastest progression from disease to a vaccine in history (Figure 3-4).
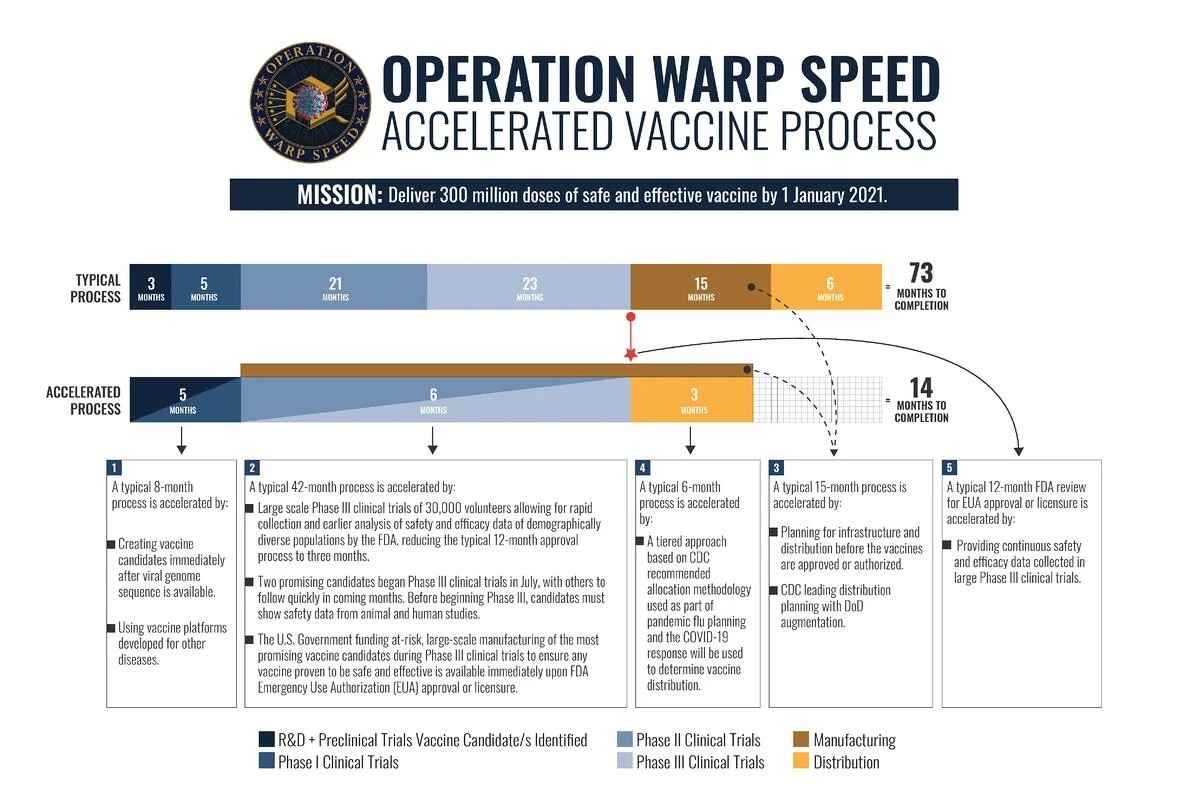
Figure 3-4: Accelerated vaccine process for Operation Warp Speed.
Reproduced from US Department of Defense. 2020. Operation Warp Speed: Accelerated Vaccine Process. Accessed October 8, 2020.
*Abbreviations: CDC, Centers for Disease Control and Prevention; DoD, Department of Defense; FDA, Food and Drug Administration; R&D, Research and development
Operation Warp Speed coordinates with multiple agencies involved in the vaccine development process, including the CDC, FDA, National Institutes of Health, and Biomedical Advanced Research and Development Authority (US Department of Health and Human Services 2020).
Vaccine Types
The objective of a vaccine is to expose the vaccine recipient’s immune system to the virus or a portion of the virus in order to trigger a response without causing the illness. Several different types of vaccines are currently under development by manufacturers throughout the world. Although it may seem counterintuitive for multiple candidates to simultaneously enter the testing phase, this practice is common and presents multiple approaches (Table 3-1).
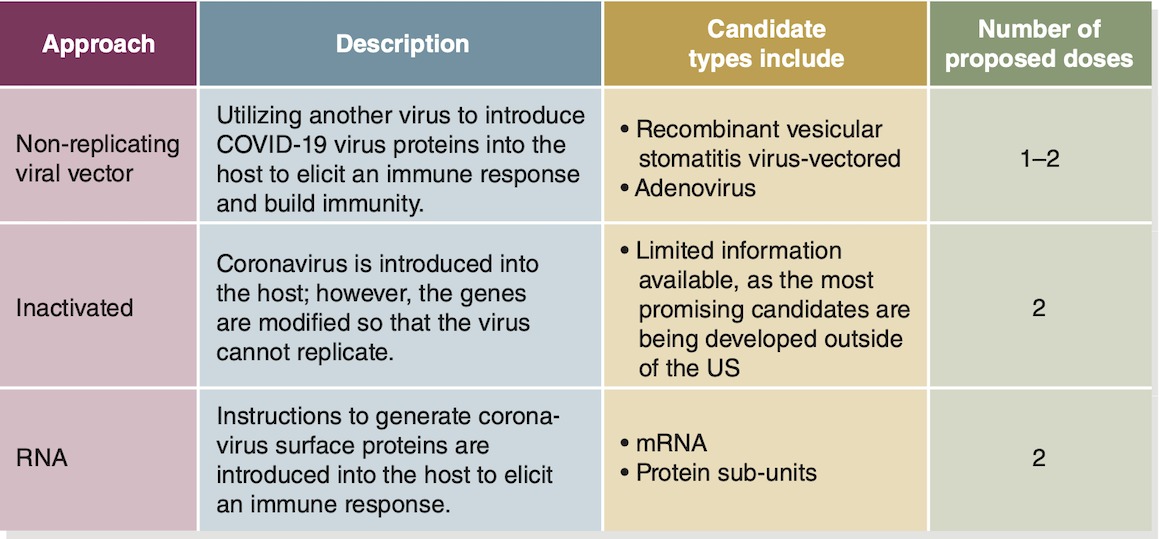
Table 3-1: Representative Approaches to a COVID-19 Vaccine
<SRC>Data from World Health Organization. Draft Landscape of COVID-19 Candidate Vaccines. Accessed October 9, 2020.
*Abbreviations: mRNA, messenger RNA; RNA, ribonucleic acid
The most likely candidates include vaccinations that introduce an altered form of the virus, introduce a surface protein found on the virus, or introduce instructions for making a protein found on the virus.
Viral- and vector-based vaccines introduce a version of the SARS-CoV-2 virus, modified to be either dead or unable to replicate, to the recipient. For example, the conventional flu vaccine delivers an inactivated virus. One type of viral COVID-19 vaccine uses the adenovirus as the virus vector. This virus otherwise causes the common cold in humans; however, when used as a vaccine vector, it does not cause illness. In fact, the adenovirus is modified to produce only a protein specific to the coronavirus, produced in a few human cells, and thus elicit a localized immune response in the recipient (Malcom 2020).
Messenger RNA (mRNA) vaccines skip the step of having the SARS-CoV-2 virus invade a human cell’s DNA. Instead, an mRNA vaccine sends instructions to the cells, encoding a message to make proteins specific to the coronavirus. Once the mRNA is taken up by a small number of the vaccine recipient’s cells, the protein is produced and the immune response targets the foreign protein. After targeting the protein, the immune system builds memory and defends the body against any virus cells that have the same surface protein (O’Callaghan, Blatz, Offit 2020).
Another notable virus-based vaccine is a recombinant vesicular stomatitis virus-vectored vaccine. It uses a virus with replication abilities to deliver antigen-producing genes from the coronavirus into human cells. The virus vector incorporates genes from the coronavirus that can be replicated in the recipient. In essence, this process creates a small amount of the virus, but with only the genes needed to recognize the coronavirus, not those that allow it to replicate. By using a virus with replication capability, in which the recipient’s immune system will respond, a more sustained immunogenic response may be produced, limiting the number of required vaccinations. This type of vaccine has also been used to develop a vaccine to the Zaire ebolavirus (O’Callaghan, Blatz, Offit 2020).
Potential Readiness
Promising vaccine candidates are progressing through clinical testing, but questions remain about whether a vaccine will be readily available by the beginning of 2021. Issues such as efficacy, side effects, adverse reactions, number of doses, availability to the general public, and length of immunity must all be weighed. In the next excerpt from Evolution of EMS: COVID-19 Guidance for EMS Providers, "Part 4: Treatment Strategies,” we’ll examine a variety of treatments and how they’ve evolved to target the distinct challenges of this virus.
References
Callaway, Ewen. 2020, August 11. “Russia’s Fast-Track Coronavirus Vaccine Draws Outrage Over Safety.” Nature. Accessed October 13, 2020.
Centers for Disease Control and Prevention. 2018, January 26. “The Journey of Your Child’s Vaccine.” Accessed October 13, 2020.
Malcom, Kelly. 2020. “The Top 5 COVID-19 Vaccine Candidates Explained.” University of Michigan. Accessed October 13, 2020.
O’Callaghan, Kevin P., Allison M. Blatz, and Paul A. Offit. 2020. “Developing a SARS-CoV-2 Vaccine at Warp Speed.” JAMA 324(5), 437-8.
O’Keefe, Shannon Mullen. 2020, August 7. “One in Three Americans Would Not Get COVID-19 Vaccine.” Gallup. Accessed October 13, 2020.
US Department of Health and Human Services. 2020, September 4. “Fact Sheet: Explaining Operation Warp Speed.” Accessed October 13, 2020.
Wee, Sui-lee, and Mariana Simões. 2020, July 16. “In Coronavirus Vaccine Race, China Strays From the Official Paths.” The New York Times. Accessed October 13, 2020.
World Health Organization. 2020. “Draft Landscape of COVID-19 Candidate Vaccines.” Accessed October 13, 2020.
3d3bb467-e9ed-4611-87cd-e46c025ac32b.webp?sfvrsn=a1b7a841_3)